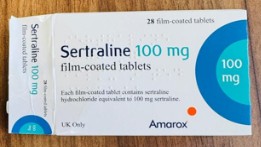
A batch of antidepressants has been recalled by health officials after packets were found to contain the wrong medication.
A patient taking Sertraline 100mg tablets found a strip of Citalopram 40mg tablets inside the sealed packet.
Sertraline and Citalopram are manufactured by the same company at the same production site, with the error believed to have occurred during secondary packaging of the blister strips in cartons.
The discovery resulted in UK-based pharmaceutical company Amarox issuing a “precautionary recall” of the batch of medication.
Officials have stressed anyone who believes they have taken Citalopram 40mg tablets by mistake should seek medical advice immediately.
 Gov.UK
Gov.UKThe Medicines and Healthcare products Regulatory Agency has also warned pharmacists to stop the supply of the affected batch and to make contact with patients who have been prescribed the product.
The MHRA stated the recall is part of measures to investigate how this occurred.
MHRA chief safety officer, Dr Alison Cave, said: “If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check the carton contains the right medication.
“You can find the batch number and expiry date printed on the side of the outer packaging.
“If the blister strips inside the carton are labelled Citalopram 40mg, please contact your pharmacy as soon as possible. If they are labelled Sertraline 100mg, no further action is needed.”
Patients who accidentally consumed Citalopram instead of or as well as Sertraline might experience headaches, nausea, mild anxiety and changes in their sleeping pattern.
Sertraline and Citalopram are both selective serotonin reuptake inhibitors (SSRIs) used to treat depression and anxiety disorders by boosting serotonin levels.
Follow STV News on WhatsApp
Scan the QR code on your mobile device for all the latest news from around the country


 Adobe Stock
Adobe Stock